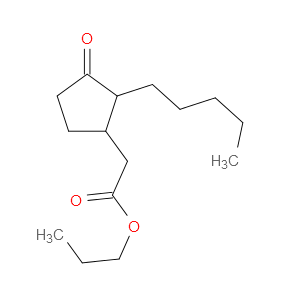
Prohydrojasmon
CAS No. 158474-72-7
Prohydrojasmon( Prohydrojasmon racemate | Propyl dihydrojasmonate )
Catalog No. M20650 CAS No. 158474-72-7
Prohydrojasmon is a synthesized plant gowth regulator which has jasmonic acid activity.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 29 | In Stock |


|
| 100MG | Get Quote | In Stock |


|
| 200MG | 33 | In Stock |


|
| 500MG | 55 | In Stock |


|
| 1G | 82 | In Stock |


|
Biological Information
-
Product NameProhydrojasmon
-
NoteResearch use only, not for human use.
-
Brief DescriptionProhydrojasmon is a synthesized plant gowth regulator which has jasmonic acid activity.
-
DescriptionProhydrojasmon is a synthesized plant gowth regulator which has jasmonic acid activity.
-
In VitroProhydrojasmon racemate (n-propyl dihydrojasmonate, PDJ) is a functional analogue of Jasmonic acid (JA) . Prohydrojasmon racemate (n-propyl dihydrojasmonate, PDJ) is a plant growth promoter.
-
In Vivo——
-
SynonymsProhydrojasmon racemate | Propyl dihydrojasmonate
-
PathwayOthers
-
TargetOther Targets
-
RecptorOthers
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number158474-72-7
-
Formula Weight254.37
-
Molecular FormulaC15H26O3
-
Purity>98% (HPLC)
-
SolubilityDMSO:54 mg/mL (212.29 mM)
-
SMILESCCCCCC1C(CC(=O)OCCC)CCC1=O
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Cohen S Barer F Itzhak I et al. Effect of prohydrojasmon on total phenolic content anthocyanin accumulation and antioxidant activity in komatsuna and lettuce[J].J Immunol Res. 2018 May 15;2018:2310970.
molnova catalog



related products
-
cholesterol-absorpti...
cholesterol-absorption inhibitor Intermediate 2 is a potent and orally active cholesterol absorption inhibitor that reduces blood cholesterol levels.
-
(Tyr27)-pTH (27-48) ...
(Tyr27)-pTH (27-48) (human)
-
Pelargonidin-3,5-O-d...
Pelargonidin-3,5-O-diglucoside chloride has antioxidant activity.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


