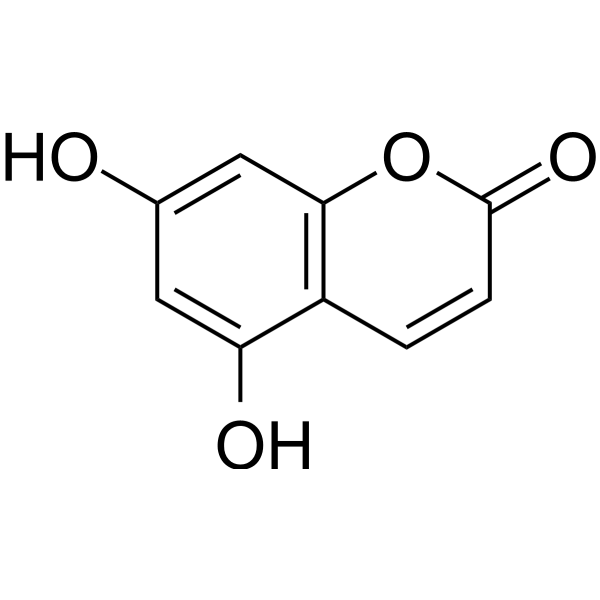
5,7-Dihydroxycoumarin
CAS No. 2732-18-5
5,7-Dihydroxycoumarin( —— )
Catalog No. M28126 CAS No. 2732-18-5
5,7-Dihydroxycoumarin is a natural product isolated from the dichloromethane extract of the inflorescences of Macaranga triloba.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 28 | In Stock |


|
| 100MG | Get Quote | In Stock |


|
| 200MG | 27 | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product Name5,7-Dihydroxycoumarin
-
NoteResearch use only, not for human use.
-
Brief Description5,7-Dihydroxycoumarin is a natural product isolated from the dichloromethane extract of the inflorescences of Macaranga triloba.
-
Description5,7-Dihydroxycoumarin is a natural product isolated from the dichloromethane extract of the inflorescences of Macaranga triloba.
-
In Vitro——
-
In Vivo——
-
Synonyms——
-
PathwayGPCR/G Protein
-
TargetAntibacterial
-
Recptorestrogen receptor
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number2732-18-5
-
Formula Weight178.143
-
Molecular FormulaC9H6O4
-
Purity>98% (HPLC)
-
SolubilityIn Vitro:?DMSO : 25 mg/mL (140.34 mM)
-
SMILESOc1cc(O)c2ccc(=O)oc2c1
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Chen SH, et al. Premarin stimulates estrogen receptor-alpha to protect against traumatic brain injury in male rats. Crit Care Med. 2009 Dec;37(12):3097-106.
molnova catalog



related products
-
Timcodar
timcodar is a non-FKBP12 binding macrolide derivative that is an efflux pump inhibitor with antimicrobial activity, inhibition of lipid accumulation, inhibition of Mycobacterium tuberculosis, and can be used in the study of obesity.
-
D13-9001
D13-9001 (D 13-9001) is a small molecule, MexAB-OprM specific efflux pump inhibitor with good potency in vitro (MPC 2 ug/mL) and displays excellent activity in vivo in a rat pneumonia model of P. aeruginosa.
-
Cefpiramide acid
Cefpiramide is a third-generation cephalosporin antibiotic.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


