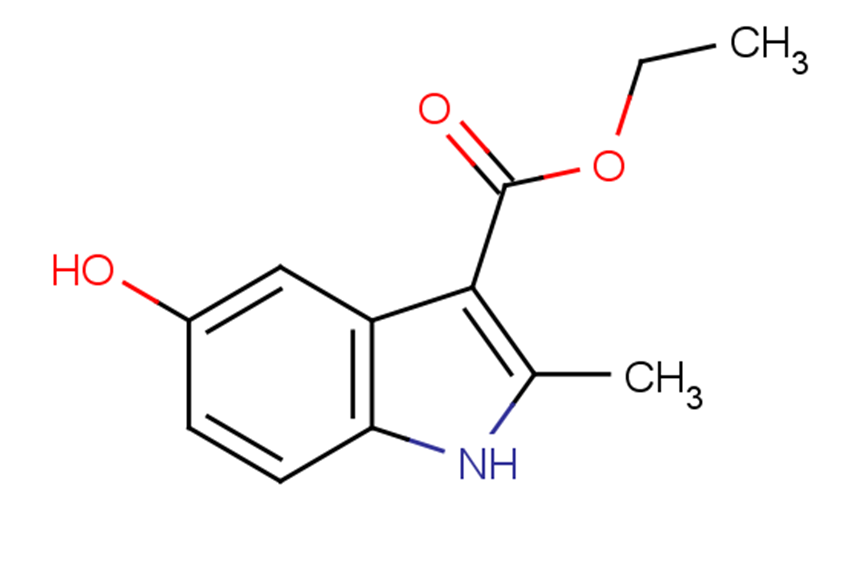
ETHYL 5-HYDROXY-2-METHYLINDOLE-3-CARBOXYLATE
CAS No. 7598-91-6
ETHYL 5-HYDROXY-2-METHYLINDOLE-3-CARBOXYLATE( —— )
Catalog No. M22206 CAS No. 7598-91-6
ETHYL 5-HYDROXY-2-METHYLINDOLE-3-CARBOXYLATE is useful for preparation and study of hypoxia-selective cytotoxicity of indolequinone antitumor agents.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 28 | In Stock |


|
| 100MG | Get Quote | In Stock |


|
| 200MG | 27 | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameETHYL 5-HYDROXY-2-METHYLINDOLE-3-CARBOXYLATE
-
NoteResearch use only, not for human use.
-
Brief DescriptionETHYL 5-HYDROXY-2-METHYLINDOLE-3-CARBOXYLATE is useful for preparation and study of hypoxia-selective cytotoxicity of indolequinone antitumor agents.
-
DescriptionETHYL 5-HYDROXY-2-METHYLINDOLE-3-CARBOXYLATE is useful for preparation and study of hypoxia-selective cytotoxicity of indolequinone antitumor agents.
-
In Vitro——
-
In Vivo——
-
Synonyms——
-
PathwayOthers
-
TargetOther Targets
-
RecptorOthers
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number7598-91-6
-
Formula Weight219.24
-
Molecular FormulaC12H13NO3
-
Purity>98% (HPLC)
-
Solubility——
-
SMILESCCOC(=O)c1c(C)nc2ccc(O)cc12
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
molnova catalog



related products
-
Methylophiopogonanon...
Methylophiopogonanone A has anti-inflammatory and anti-oxidative properties.
-
Palladium (II) aceta...
Palladium (II) acetate is a catalyst that catalyzes aromatic substitution reactions.
-
β-Endorphin (6-31), ...
β-Endorphin, an endogenous opioid neuropeptide, is an opioid receptor agonist. β-Endorphin binds preferentially to μ-opioid receptors and is produced in certain neurons of the central and peripheral nervous system and is one of three endorphins produced in humans. β-Endorphin can be used to reduce stress and maintain homeostasis in the body and is involved in neurological pain perception regulation.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


