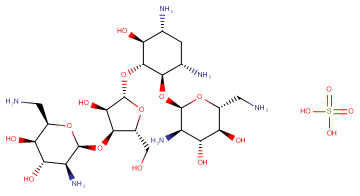
Neomycin Sulphate B
CAS No. 4146-30-9
Neomycin Sulphate B ( Framycetin sulphate | Neomycin sulphate B )
Catalog No. M14419 CAS No. 4146-30-9
Neomycin belongs to aminoglycoside class of antibiotics that contain two or more aminosugars connected by glycosidic bonds.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 25MG | 37 | In Stock |


|
| 50MG | 60 | In Stock |


|
| 100MG | 83 | In Stock |


|
| 200MG | 119 | In Stock |


|
| 500MG | 204 | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameNeomycin Sulphate B
-
NoteResearch use only, not for human use.
-
Brief DescriptionNeomycin belongs to aminoglycoside class of antibiotics that contain two or more aminosugars connected by glycosidic bonds.
-
DescriptionNeomycin belongs to aminoglycoside class of antibiotics that contain two or more aminosugars connected by glycosidic bonds. (In Vitro):The inhibition of RNase P RNA cleavage by Framycetin sulfate (Neomycin Bsulfate; Fradiomycin Bsulfate) is sensitive to pH and an increase in pH suppresses the inhibition in other systems.Framycetin sulfate targets the bacterial and human ribosome and affect translation. 5″-azido neomycin B and Framycetin sulfate selectively inhibit production of the mature miRNA, boosts a downstream protein, and inhibits invasion in HCC cell line.Framycetin sulfate binds to a structural rather than a sequence motif of the RNA. Its primary cognate target is the decoding site of the 16S rRNA, but it also binds to the Rev-responsive element in HIV-1, group I introns, and the hammerhead ribozyme, and thus inhibits their biological function.Framycetin sulfate induces misreading of the genetic code during translation and inhibits several ribozymes. The ribosomal target site is the 16 S rRNA 1400 to 1500 region.
-
In Vitro——
-
In Vivo——
-
SynonymsFramycetin sulphate | Neomycin sulphate B
-
PathwayMicrobiology/Virology
-
TargetAntibiotic
-
RecptorAntibiotic
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number4146-30-9
-
Formula Weight712.22
-
Molecular FormulaC23H48N6O17S
-
Purity>98% (HPLC)
-
SolubilityDMSO: 10 mM
-
SMILESC1[C@H]([C@@H]([C@H]([C@@H]([C@H]1N)O[C@@H]2[C@@H]([C@H]([C@@H]([C@H](O2)CN)O)O)N)O[C@H]3[C@@H]([C@@H]([C@H](O3)CO)O[C@H]4[C@H]([C@@H]([C@H]([C@H](O4)CN)O)O)N)O)O)N.OS(=O)(=O)O
-
Chemical Name(2R,3R,4S,5S,6S)-5-amino-2-(aminomethyl)-6-(((2R,3S,4R,5S)-5-(((1R,2R,3S,5R,6S)-3,5-diamino-2-(((2R,3R,4R,5S,6R)-3-amino-6-(aminomethyl)-4,5-dihydroxytetrahydro-2H-pyran-2-yl)oxy)-6-hydroxycyclohexyl)oxy)-4-hydroxy-2-(hydroxymethyl)tetrahydrofuran-3-yl)oxy)tetrahydro-2H-pyran-3,4-diol sulfate
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
molnova catalog



related products
-
2-O-(2-Azidoethyl)ad...
2'-O-(2-azidoethyl)adenosine is a purine nucleoside analog with potential antitumor activity.
-
Gentamicin
Gentamicin (Centicin) is a broad-spectrum aminoglycoside antibiotic with antibacterial activity, inhibiting the growth of Gram-positive and Gram-negative bacteria, and can be used to study ocular infections.
-
Cefuzonam
Cefuzonam is a novel cephalosporin with anti-inflammatory and antibacterial activity that can be used in studies of respiratory tract infections, tonsillitis, and hepatobiliary infections.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


