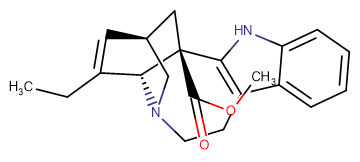
Catharanthine
CAS No. 2468-21-5
Catharanthine ( (+)-3,4-Didehydrocoronaridine )
Catalog No. M13718 CAS No. 2468-21-5
Catharanthine inhibits nicotinic receptor mediated diaphragm contractions with IC50 of 59.6 μM.
Purity : >98%(HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 50MG | 35 | In Stock |


|
| 100MG | 51 | In Stock |


|
| 200MG | Get Quote | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameCatharanthine
-
NoteResearch use only, not for human use.
-
Brief DescriptionCatharanthine inhibits nicotinic receptor mediated diaphragm contractions with IC50 of 59.6 μM.
-
DescriptionCatharanthine inhibits nicotinic receptor mediated diaphragm contractions with IC50 of 59.6 μM.
-
Synonyms(+)-3,4-Didehydrocoronaridine
-
PathwayEndocrinology/Hormones
-
TargetAChR
-
RecptornAChR
-
Research AreaNeurological Disease
-
Indication——
Chemical Information
-
CAS Number2468-21-5
-
Formula Weight336.42
-
Molecular FormulaC21H24N2O2
-
Purity>98%(HPLC)
-
SolubilityEthanol: 3 mg/mL (8.91 mM); DMSO: 67 mg/mL (199.14 mM)
-
SMILESCCC1=C[C@H]2C[C@]3([C@@H]1N(C2)CCC4=C3NC5=CC=CC=C45)C(=O)OC
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Pereira DM, et al. Phytomedicine, 2010, 17(8-9), 646-652.
molnova catalog


related products
-
SEN 12333
α7 nicotinic acetylcholine receptor (nAChR) agonist.
-
Encenicline
Encenicline is a selective α7 nicotinic acetylcholine receptor?(nAChRs) agonist in development for treating cognitive impairment in schizophrenia and Alzheimer's disease.
-
Catestatin
Non-competitive nicotinic cholinergic antagonist; selectively inhibits nicotinic-stimulated catecholamine secretion from chromaffin cells and noradrenergic neurons (IC50 ~ 200 nM). Blocks nicotinic-induced cationic signaling (IC50 ~ 200 - 250 nM) and inhibits nicotinic-agonist induced desensitization of catecholamine release. Also stimulates mast cell release of histamine via a separate mechanism.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


