Tenatoprazole
CAS No. 113712-98-4
Tenatoprazole( TU-199 | Tenatoprazole | Ulsacare | Protop )
Catalog No. M17835 CAS No. 113712-98-4
Tenatoprazole, a prodrug of the proton pump inhibitor (PPI) class, can inhibit proton transport (IC50: 3.2 μM).
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 10MG | 42 | In Stock |


|
| 25MG | 57 | In Stock |


|
| 50MG | 68 | In Stock |


|
| 100MG | 96 | In Stock |


|
| 200MG | 147 | In Stock |


|
| 500MG | 250 | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameTenatoprazole
-
NoteResearch use only, not for human use.
-
Brief DescriptionTenatoprazole, a prodrug of the proton pump inhibitor (PPI) class, can inhibit proton transport (IC50: 3.2 μM).
-
DescriptionTenatoprazole, also known as TU-199, is a proton pump inhibitor drug candidate that was undergoing clinical testing as a potential treatment for reflux oesophagitis and peptic ulcer. Tenatoprazole has an imidazopyridine ring in place of the benzimidazole moiety found in other proton pump inhibitors, and has a half-life about seven times longer than other PPIs.
-
In Vitro——
-
In Vivo——
-
SynonymsTU-199 | Tenatoprazole | Ulsacare | Protop
-
PathwayOthers
-
TargetOther Targets
-
RecptorProton pump
-
Research AreaInflammation/Immunology
-
Indication——
Chemical Information
-
CAS Number113712-98-4
-
Formula Weight346.4
-
Molecular FormulaC16H18N4O3S
-
Purity>98% (HPLC)
-
SolubilityIn Vitro:?DMSO : 50 mg/mL (144.34 mM)
-
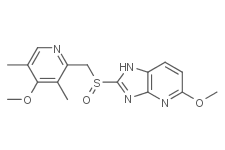
SMILESCOc1ccc2[nH]c(nc2n1)S(=O)Cc1c(C)c(OC)c(C)cn1
-
Chemical Name5-methoxy-2-[(4-methoxy-3,5-dimethylpyridin-2-yl)methylsulfinyl]-1H-imidazo[4,5-b]pyridine
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1. Shin JM, et al. Biochem Pharmacol, 2006, 71(6), 837-849.
molnova catalog



related products
-
Thiohexam
Thiohexam is a rubber vulcanization accelerator.
-
PS 48
PS 48 has been shown to be a PKB Kinase (PDK1) activator (Kd: 10.3 μM). This compound selectively binds to the PIF-binding pocket of PKB Kinase (PDK1).
-
Baldrinal
Baldrinal, valtrate, and acevaltrate are the quality control compounds of Valeriana jatamansi Jones.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


