Spiperone
CAS No. 749-02-0
Spiperone( Spiperone | E 525 | E-525 | E525 | R 5147 | R-5147 | R5147 | NSC 170983 )
Catalog No. M15854 CAS No. 749-02-0
A spiro butyrophenone analog similar to HALOPERIDOL and other related compounds. It has been recommended in the treatment of SCHIZOPHRENIA.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 5MG | 35 | In Stock |


|
| 10MG | 52 | In Stock |


|
| 100MG | Get Quote | In Stock |


|
| 200MG | Get Quote | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameSpiperone
-
NoteResearch use only, not for human use.
-
Brief DescriptionA spiro butyrophenone analog similar to HALOPERIDOL and other related compounds. It has been recommended in the treatment of SCHIZOPHRENIA.
-
DescriptionA spiro butyrophenone analog similar to HALOPERIDOL and other related compounds. It has been recommended in the treatment of schizophrenia.
-
In Vitro——
-
In Vivo——
-
SynonymsSpiperone | E 525 | E-525 | E525 | R 5147 | R-5147 | R5147 | NSC 170983
-
PathwayGPCR/G Protein
-
TargetDopamine Receptor
-
RecptorDopamine
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number749-02-0
-
Formula Weight395.47
-
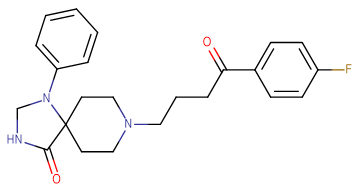
Molecular FormulaC23H26FN3O2
-
Purity>98% (HPLC)
-
SolubilityDMSO: 10 mM
-
SMILESO=C1NCN(C2=CC=CC=C2)C13CCN(CCCC(C4=CC=C(F)C=C4)=O)CC3
-
Chemical Name8-(4-(4-fluorophenyl)-4-oxobutyl)-1-phenyl-1,3,8-triazaspiro[4.5]decan-4-one
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Zheng LT, et al.J Neurochem. 2008 Dec;107(5):1225-35.
molnova catalog



related products
-
Chlorpromazine
Chlorpromazine (CPZ) is a phenothiazine acting as dopamine antagonist.Chlorpromazine is a low-potency typical antipsychotic agent for the treatment of psychotic disorders such as schizophrenia.Chlorpromazine (CPZ) is a phenothiazine acting as dopamine antagonist.?
-
FIIN-2
FIIN-2, an irreversible, pan-FGFR inhibitor, inhibits FGFR1/2/3/4 with IC50 of 3.09 nM, 4.3 nM, 27 nM and 45.3 nM, respectively.
-
SLV-310
SLV-310 is a potent dopamine D2 receptor antagonist and 5-HT reuptake receptor inhibitor used in the study of neurological disorders such as bipolar disorder and schizophrenia.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


