Sirt2-PROTAC-1
CAS No. 2098487-75-1
Sirt2-PROTAC-1( Sirt2-PROTAC-1 | Sirt2 PROTAC-1 | Sirt2-PROTAC1 )
Catalog No. M13316 CAS No. 2098487-75-1
A novel SirReal-based PROTAC that induces isotype-selective Sirt2 degradation (IC50=0.25 uM).
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 100MG | Get Quote | Get Quote |


|
| 200MG | Get Quote | Get Quote |


|
| 500MG | Get Quote | Get Quote |


|
| 1G | Get Quote | Get Quote |


|
Biological Information
-
Product NameSirt2-PROTAC-1
-
NoteResearch use only, not for human use.
-
Brief DescriptionA novel SirReal-based PROTAC that induces isotype-selective Sirt2 degradation (IC50=0.25 uM).
-
DescriptionA novel SirReal-based PROTAC that induces isotype-selective Sirt2 degradation (IC50=0.25 uM).
-
In VitroWestern Blot Analysis Cell Line:HeLa cells Concentration:10 μM Incubation Time:1-6 hours Result:Caused Sirt2 degradation, but showed no effect on Sirt1 levels.
-
In Vivo——
-
SynonymsSirt2-PROTAC-1 | Sirt2 PROTAC-1 | Sirt2-PROTAC1
-
PathwayPROTACs
-
TargetPROTAC
-
RecptorPROTAC
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number2098487-75-1
-
Formula Weight852.942
-
Molecular FormulaC40H40N10O8S2
-
Purity>98% (HPLC)
-
SolubilityDMSO : ≥ 100 mg/mL 117.24 mM; H2O : < 0.1 mg/mL
-
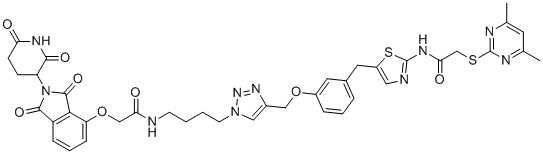
SMILESO=C(NC1=NC=C(CC2=CC=CC(OCC3=CN(CCCCNC(COC4=CC=CC(C(N5C(CC6)C(NC6=O)=O)=O)=C4C5=O)=O)N=N3)=C2)S1)CSC7=NC(C)=CC(C)=N7
-
Chemical Name2-((4,6-dimethylpyrimidin-2-yl)thio)-N-(5-(3-((1-(4-(2-((2-(2,6-dioxopiperidin-3-yl)-1,3-dioxoisoindolin-4-yl)oxy)acetamido)butyl)-1H-1,2,3-triazol-4-yl)methoxy)benzyl)thiazol-2-yl)acetamide
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1. Schiedel M, et al. J Med Chem. 2017 Apr 17. doi: 10.1021/acs.jmedchem.6b01872.
molnova catalog



related products
-
dBET1
dBET1 is a proteolysis-targeting chimera (PROTAC) molecule that appendes a competitive antagonist of BET bromodomain (JQ1) to a phthalimide moiety to hijack the cereblon E3 ubiquitin ligase complex.
-
Ac4ManNAz
Ac4ManNAz is a PEG-based PROTAC linker that can be used in the synthesis of PROTACs.
-
MT-802
MT-802 (MT802) is a potent BTK PROTAC that induces degradation of both wild-type and C481S mutant BTK (DC50=9.1 nM).



 Cart
Cart
 sales@molnova.com
sales@molnova.com


