SBI-425
CAS No. 1451272-71-1
SBI-425( —— )
Catalog No. M26818 CAS No. 1451272-71-1
SBI-425 is an effective and orally bioavailable inhibitor of tissue-nonspecific alkaline phosphatase (TNAP).
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 5MG | 65 | Get Quote |


|
| 10MG | 107 | Get Quote |


|
| 25MG | 173 | Get Quote |


|
| 50MG | 252 | Get Quote |


|
| 100MG | 374 | Get Quote |


|
| 200MG | Get Quote | Get Quote |


|
| 500MG | Get Quote | Get Quote |


|
| 1G | Get Quote | Get Quote |


|
Biological Information
-
Product NameSBI-425
-
NoteResearch use only, not for human use.
-
Brief DescriptionSBI-425 is an effective and orally bioavailable inhibitor of tissue-nonspecific alkaline phosphatase (TNAP).
-
DescriptionSBI-425 is an effective and orally bioavailable inhibitor of tissue-nonspecific alkaline phosphatase (TNAP). SBI-425 effectively inhibits TNAP in the vasculature and improves cardiovascular parameters and survival without a detectable change in the bone.
-
In VitroSBI-425 treatment suppresses Foxp3 expression in CD4+ and CD8+ T cells in splenocytes from CLP-injured mice.
-
In Vivo——
-
Synonyms——
-
PathwayOthers
-
TargetOther Targets
-
Recptor——
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number1451272-71-1
-
Formula Weight341.77
-
Molecular FormulaC13H12ClN3O4S
-
Purity>98% (HPLC)
-
SolubilityIn Vitro:?DMSO : 86.67 mg/mL (253.59 mM)
-
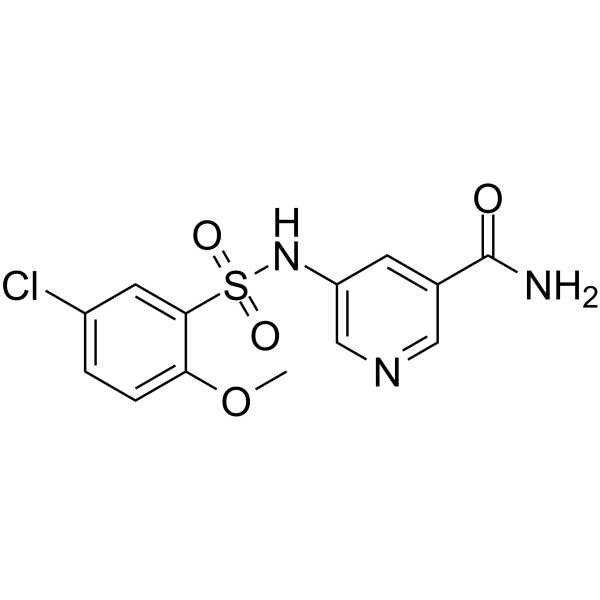
SMILESCOc1ccc(Cl)cc1S(=O)(=O)Nc1cncc(c1)C(N)=O
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.A L Tarentino, et al. 2-Iminothiolane: a reagent for the introduction of sulphydryl groups into oligosaccharides derived from asparagine-linked glycans. Glycobiology. 1993 Jun;3(3):279-85.
molnova catalog



related products
-
Anthracene-9-carboxy...
Anthracene-9-carboxylic acid is an inhibitor of chloride transport with a moderate to strong inhibitory action on PKA activated cardiac IcI.
-
Bixin
Bixin is isolated from the seeds of Bixa orellana and possesses anti-tumor, anti-inflammatory, and antioxidant activities.
-
Salvianan
Salviane exhibits the most potent cytotoxicity with a CD50 range of 30.4-39.5 microM against HeLa (cervical epitheloid carcinoma), HepG2 (hepatocellular carcinoma), and OVCAR-3 (ovarian adenocarcinoma) cell lines in a dose-dependent manner.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


