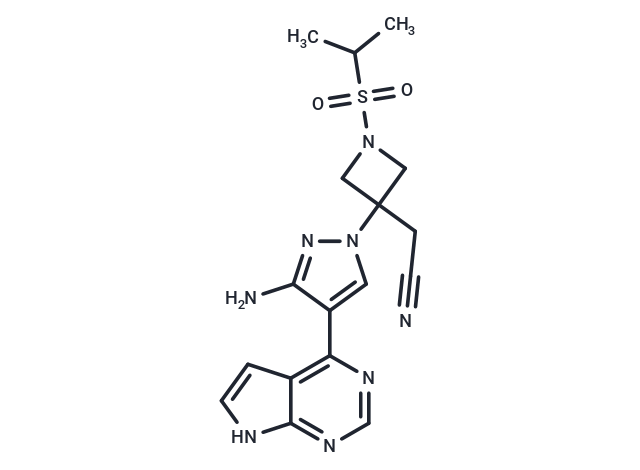
Pumecitinib
CAS No. 2401057-12-1
Pumecitinib( —— )
Catalog No. M35964 CAS No. 2401057-12-1
Pumecitinib is a Janus kinase (JAK) inhibitor. Pumecitinib exhibits anti-inflammatory activity.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 50 | In Stock |


|
| 5MG | 42 | In Stock |


|
| 10MG | 67 | In Stock |


|
| 25MG | 124 | In Stock |


|
| 50MG | 193 | In Stock |


|
| 100MG | 275 | In Stock |


|
| 200MG | Get Quote | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NamePumecitinib
-
NoteResearch use only, not for human use.
-
Brief DescriptionPumecitinib is a Janus kinase (JAK) inhibitor. Pumecitinib exhibits anti-inflammatory activity.
-
DescriptionPumecitinib is a Janus kinase (JAK) inhibitor with anti-inflammatory activity.
-
In Vitro——
-
In Vivo——
-
Synonyms——
-
PathwayAngiogenesis
-
TargetJAK
-
RecptorJAK
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number2401057-12-1
-
Formula Weight400.46
-
Molecular FormulaC17H20N8O2S
-
Purity>98% (HPLC)
-
SolubilityIn Vitro:?DMSO : 125 mg/mL (312.14 mM; Ultrasonic (<60°C)
-
SMILESCC(C)S(=O)(=O)N1CC(CC#N)(C1)n1cc(c(N)n1)-c1ncnc2[nH]ccc12
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1. WHO Drug Information-World Health Organization (WHO).
molnova catalog



related products
-
Lorpucitinib
Lorpucitinib (JNJ-64251330) is an orally available, selective and potent JAK kinase inhibitor for the study of inflammatory and gastrointestinal disorders associated with Janus kinase (JAK) signaling.
-
SHR0302
SHR0302 is a JAK inhibitor that binds JAK1 with stronger affinity than others (Selectivity for JAK1 is more than 10 times for JAK2, 77 times for JAK3, 420 times for Tyk2).
-
Dracorhodin perochlo...
Dracorhodin perchlorate inhibits cell growth, and induces apoptosis in fibroblasts in a dose-and time-dependent manner, arresting cell cycle at G1 phase, may as a candidate for anti-breast cancer.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


