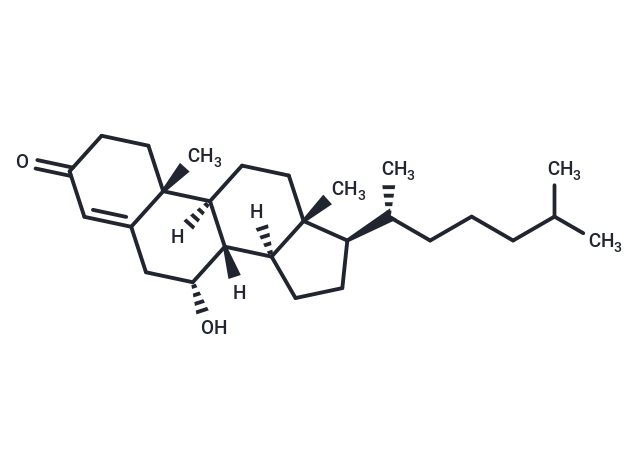
7α-Hydroxy-4-cholesten-3-one
CAS No. 3862-25-7
7α-Hydroxy-4-cholesten-3-one( —— )
Catalog No. M32760 CAS No. 3862-25-7
7α-Hydroxy-4-cholesten-3-one is an intermediate in bile acid synthesis from cholesterol, a PXR agonist, and a biomarker for bile acid loss and diseases related to defective bile acid biosynthesis.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 5MG | 581 | In Stock |


|
| 10MG | 847 | In Stock |


|
| 100MG | Get Quote | In Stock |


|
| 200MG | Get Quote | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product Name7α-Hydroxy-4-cholesten-3-one
-
NoteResearch use only, not for human use.
-
Brief Description7α-Hydroxy-4-cholesten-3-one is an intermediate in bile acid synthesis from cholesterol, a PXR agonist, and a biomarker for bile acid loss and diseases related to defective bile acid biosynthesis.
-
Description7α-Hydroxy-4-cholesten-3-one is an intermediate in synthesis of bile acids from cholesterol. 7α-Hydroxy-4-cholesten-3-one is a pregnane X receptor (PXR) agonist. 7α-Hydroxy-cholest-4-en-3-one is a biomarker for bile acid loss, irritable bowel syndrome, and other diseases associated with defective bile acid biosynthesis. 7α-Hydroxy-cholest-4-en-3-one is the physiological substrate for CYP8B1.
-
In Vitro7α-Hydroxy-4-cholesten-3-one is found relatively upstream in the biosynthetic pathway to bile acids (e.g. chenodeoxycholic acid). The first step is the incorporation of the 7α-hydroxy group onto cholesterol by cytochrome P450 7A1, and the second step is the oxidation and isomerization of the 3-hydroxy group and the Δ5,6-double bond by 3β-hydroxy steroid dehydrogenase to yield 7α-Hydroxy-4-cholesten-3-one. The deletion of the gene that expresses P450 27A1, which is found downstream in the bile acid pathway, results in the accumulation of the precursor, 7α-Hydroxy-4-cholesten-3-one.
-
In Vivo7α-Hydroxy-4-cholesten-3-one strongly relates to the hepatic enzymatic activity of CYP7A1 at steady-state conditions as well as during the rapid diurnal changes that occur in the rat. That serum 7α-Hydroxy-4-cholesten-3-one has a pronounced diurnal rhythm.
-
Synonyms——
-
PathwayOthers
-
TargetOther Targets
-
RecptorOthers
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number3862-25-7
-
Formula Weight400.64
-
Molecular FormulaC27H44O2
-
Purity>98% (HPLC)
-
SolubilityIn Vitro:?DMSO : 100 mg/mL (249.60 mM; Ultrasonic )
-
SMILES[H][C@@]12CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])[C@H](O)CC2=CC(=O)CC[C@]12C
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1. Offei SD, et al. Chemical synthesis of 7α-hydroxycholest-4-en-3-one, a biomarker for irritable bowel syndrome and bile acid malabsorption. Steroids. 2019 Nov;151:108449.?
molnova catalog



related products
-
Isokadsuranin
Isokadsuranin is a natural product from Kadsura coccinea.
-
Rhodopsin Epitope Ta...
Rhodopsin Epitope Tag is a 9-amino acid peptide representing C terminus of bovine rhodopsin widely used as an epitope tag. A number of anti-rhodopsin antibodies can recognize this epitope.
-
Baccatin III
Baccatin III is an isolate of the Pacific yew tree (Taxus brevifolia) and related species. Baccatin III is a precursor to the anti-Y drug paclitaxel (Taxol).



 Cart
Cart
 sales@molnova.com
sales@molnova.com


