Ferulamide
CAS No. 61012-31-5
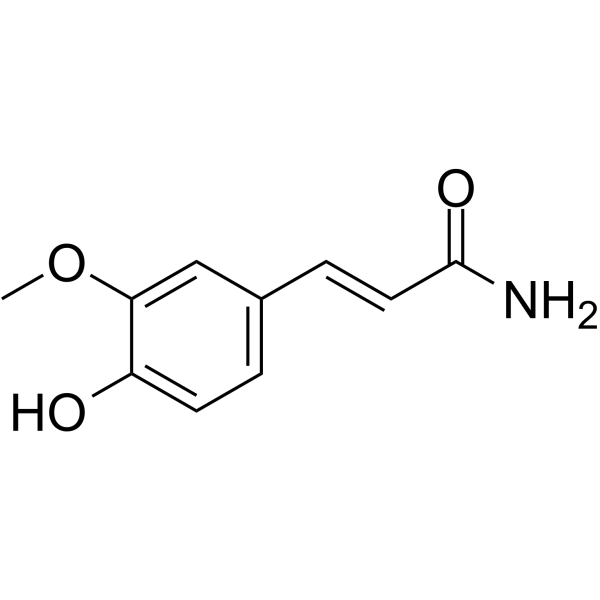
Ferulamide( 4-Hydroxy-3-methoxycinnamide )
Catalog No. M29143 CAS No. 61012-31-5
Ferulamide, a Ferulic acid derivative, shows potent inhibitory activity against arachidonic acid-induced platelet aggregation.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 371 | In Stock |


|
| 5MG | 432 | In Stock |


|
| 10MG | 636 | In Stock |


|
| 25MG | 998 | In Stock |


|
| 50MG | 1321 | In Stock |


|
| 100MG | 1786 | In Stock |


|
| 200MG | Get Quote | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameFerulamide
-
NoteResearch use only, not for human use.
-
Brief DescriptionFerulamide, a Ferulic acid derivative, shows potent inhibitory activity against arachidonic acid-induced platelet aggregation.
-
DescriptionFerulamide, a Ferulic acid derivative, shows potent inhibitory activity against arachidonic acid-induced platelet aggregation.(In Vitro):A series of Ferulamide derivatives were prepared and evaluated for their anti-platelet activities. Some of these compounds showed potent inhibitory activity against arachidonic acid-induced platelet aggregation. Their structure-activity relationships are also discussed.
-
In Vitro——
-
In Vivo——
-
Synonyms4-Hydroxy-3-methoxycinnamide
-
PathwayOthers
-
TargetOther Targets
-
Recptor——
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number61012-31-5
-
Formula Weight193.202
-
Molecular FormulaC10H11NO3
-
Purity>98% (HPLC)
-
Solubility——
-
SMILESCOc(cc(/C=C/C(N)=O)cc1)c1O
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
molnova catalog



related products
-
Tyrphostin 9
Tyrphostin 9 is an Agricultural acaricide, now superseded. Tyrphostin 9 is firstly designed as an EGFR inhibitor
-
(D-Pro4,D-Trp7·9·10,...
(D-Pro4,D-Trp7·9·10,Val8)-Substance P (4-11)
-
3'-Deoxyguanosine
3'-Deoxyguanosine is a ligand that can be complexed with enzymes such as purine nucleoside phosphorylase and receptors.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


