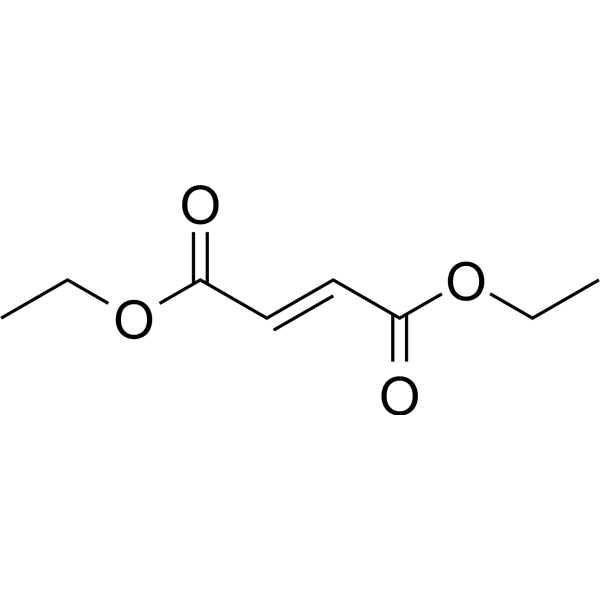
Diethyl fumarate
CAS No. 623-91-6
Diethyl fumarate( —— )
Catalog No. M28328 CAS No. 623-91-6
Diethyl fumarate is a decomposition product of the pesticide Malathion. Diethyl fumarate causes nonimmune contact urticaria on the skin.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 28 | In Stock |


|
| 100MG | Get Quote | In Stock |


|
| 200MG | Get Quote | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | 27 | In Stock |


|
Biological Information
-
Product NameDiethyl fumarate
-
NoteResearch use only, not for human use.
-
Brief DescriptionDiethyl fumarate is a decomposition product of the pesticide Malathion. Diethyl fumarate causes nonimmune contact urticaria on the skin.
-
DescriptionDiethyl fumarate is a decomposition product of the pesticide Malathion. Diethyl fumarate causes nonimmune contact urticaria on the skin.
-
In Vitro——
-
In Vivo——
-
Synonyms——
-
PathwayOthers
-
TargetOther Targets
-
Recptor——
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number623-91-6
-
Formula Weight172.18
-
Molecular FormulaC8H12O4
-
Purity>98% (HPLC)
-
SolubilityIn Vitro:?DMSO : 140 mg/mL (813.10 mM)
-
SMILESCCOC(=O)\C=C\C(=O)OCC
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Kurohane K, et al. An Aliphatic Ester Diisopropyl Sebacate Exhibited an Adjuvant Effect on Fluorescein Isothiocyanate-Induced Contact Hypersensitivity Mouse Models. Biol Pharm Bull. 2018 Jan 1;41(1):147-150.
molnova catalog



related products
-
Grandifloroside-3-gl...
Grandifloroside-3''-glucoside
-
6-ROX
6-ROX is a fluorescent oligonucleotide marker and acts as an acceptor molecule coupled to 5-FAM as the donor in FRET imaging with excitation of 568nm and emission of 568nm.
-
Dodecylphosphocholin...
Dodecylphosphocholine is a substrate-type inhibitor of pancreas phospholipase A2. Dodecylphosphocholine is a detergent.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


