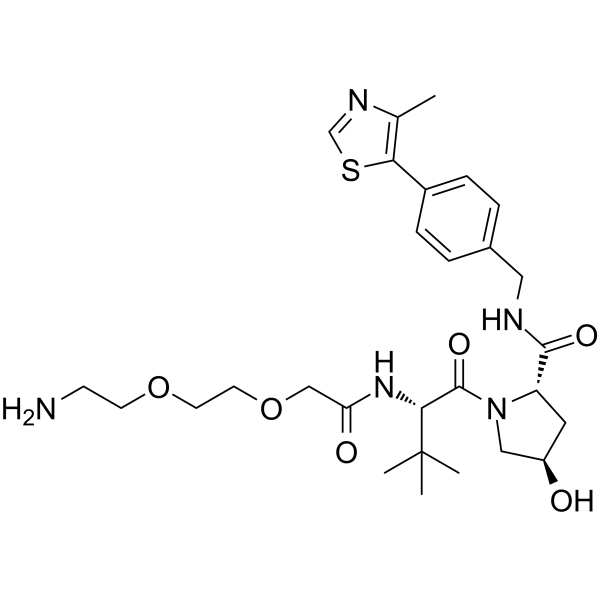
(S,R,S)-AHPC-PEG2-NH2
CAS No. 2010159-60-9
(S,R,S)-AHPC-PEG2-NH2( VH032-PEG2-NH2 | VHL Ligand-Linker Conjugates 3 | E3 ligase Ligand-Linker Conjugates 6 Free Base )
Catalog No. M26953 CAS No. 2010159-60-9
(S,R,S)-AHPC-PEG2-NH2 (VH032-PEG2-NH2) is a synthesized E3 ligase ligand-linker conjugate that incorporates the (S,R,S)-AHPC based VHL ligand and 2-unit PEG linker used in the synthesis of PROTACs.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 100MG | 241 | In Stock |


|
| 200MG | 376 | In Stock |


|
| 500MG | 896 | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product Name(S,R,S)-AHPC-PEG2-NH2
-
NoteResearch use only, not for human use.
-
Brief Description(S,R,S)-AHPC-PEG2-NH2 (VH032-PEG2-NH2) is a synthesized E3 ligase ligand-linker conjugate that incorporates the (S,R,S)-AHPC based VHL ligand and 2-unit PEG linker used in the synthesis of PROTACs.
-
Description(S,R,S)-AHPC-PEG2-NH2 (VH032-PEG2-NH2) is a synthesized E3 ligase ligand-linker conjugate that incorporates the (S,R,S)-AHPC based VHL ligand and 2-unit PEG linker used in the synthesis of PROTACs.(In Vitro):PROTACs contain two different ligands connected by a linker; one is a ligand for an E3 ubiquitin ligase and the other is for the target protein. PROTACs exploit the intracellular ubiquitin-proteasome system to selectively degrade target proteins.
-
In VitroPROTACs contain two different ligands connected by a linker; one is a ligand for an E3 ubiquitin ligase and the other is for the target protein. PROTACs exploit the intracellular ubiquitin-proteasome system to selectively degrade target proteins.
-
In Vivo——
-
SynonymsVH032-PEG2-NH2 | VHL Ligand-Linker Conjugates 3 | E3 ligase Ligand-Linker Conjugates 6 Free Base
-
PathwayOthers
-
TargetOther Targets
-
Recptorσ1
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number2010159-60-9
-
Formula Weight575.72
-
Molecular FormulaC28H41N5O6S
-
Purity>98% (HPLC)
-
Solubility——
-
SMILESO=C([C@H]1N(C([C@@H](NC(COCCOCCN)=O)C(C)(C)C)=O)C[C@H](O)C1)NCC2=CC=C(C3=C(C)N=CS3)C=C2
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Lan Y, et al. Synthesis and biological evaluation of novel sigma-1 receptor antagonists based on pyrimidine scaffold as agents for treating neuropathic pain. J Med Chem. 2014 Dec 26;57(24):10404-23.
molnova catalog



related products
-
Chiniofon
Chiniofon, a halogenated 8-hydroxyquinoline, is used as an antiamebic drug. This type of anti-amebic drug exerts an anti-amebic effect by inhibiting intestinal commensal bacteria and affects amoebic dysentery, but has no effect on extra-intestinal amoebic parasites.
-
Gardenin D
Gardenin D has antioxidant, and antiproliferative activities.
-
CEF10
CEF10



 Cart
Cart
 sales@molnova.com
sales@molnova.com


