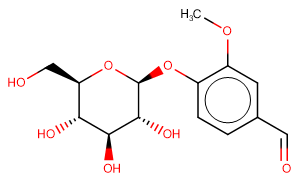
Glucovanillin
CAS No. 494-08-6
Glucovanillin( Vanillin 4-O-β-D-Glucoside )
Catalog No. M21276 CAS No. 494-08-6
Glucovanillin belongs to the class of organic compounds known as phenolic glycosides.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 1 mL x 10 mM in DMSO | 28 | In Stock |


|
| 10MG | 29 | In Stock |


|
| 25MG | 61 | In Stock |


|
| 50MG | 99 | In Stock |


|
| 100MG | 157 | In Stock |


|
| 200MG | 235 | In Stock |


|
| 500MG | Get Quote | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameGlucovanillin
-
NoteResearch use only, not for human use.
-
Brief DescriptionGlucovanillin belongs to the class of organic compounds known as phenolic glycosides.
-
DescriptionGlucovanillin belongs to the class of organic compounds known as phenolic glycosides.
-
In Vitro——
-
In Vivo——
-
SynonymsVanillin 4-O-β-D-Glucoside
-
PathwayOthers
-
TargetOther Targets
-
RecptorOthers
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number494-08-6
-
Formula Weight314.29
-
Molecular FormulaC14H18O8
-
Purity>98% (HPLC)
-
SolubilityIn Vitro:?DMSO : 100 mg/mL (318.18 mM)
-
SMILESCOc(cc(C=O)cc1)c1O[C@@H]([C@@H]([C@H]1O)O)O[C@H](CO)[C@H]1O
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Chen Y Gu F Li J et al. Involvement of Colonizing Bacillus Isolates in Glucovanillin Hydrolysis during the Curing of Vanilla planifolia Andrews[J]. Applied & Environmental Microbiology 2015 81(15):4947-4954.
molnova catalog



related products
-
Z-YVAD-FMK
AA-Z-YVAD-FMK is a Irreversible caspase-1 inhibitor.
-
D-Glucosaminic acid
D-Glucosaminic Acid is a useful starting material for the synthesis of aldonic acids.
-
D-Lactose monohydra...
Lactose is the major sugar present in milk and the main source of energy supplied to the newborn mammalian in its mother's milk.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


