LY-354740
CAS No. 176199-48-7
LY-354740( Eglumegad | Eglumetad )
Catalog No. M12658 CAS No. 176199-48-7
A potent and selective group 2 mGluRs agonist with EC50s of 55 ± 17 nM; possesses no activity at iGluR or group 1 mGluR (>100 uM).
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 200MG | 1125 | Get Quote |


|
| 500MG | Get Quote | Get Quote |


|
| 1G | Get Quote | Get Quote |


|
Biological Information
-
Product NameLY-354740
-
NoteResearch use only, not for human use.
-
Brief DescriptionA potent and selective group 2 mGluRs agonist with EC50s of 55 ± 17 nM; possesses no activity at iGluR or group 1 mGluR (>100 uM).
-
DescriptionA potent and selective group 2 mGluRs agonist with EC50s of 55 ± 17 nM; possesses no activity at iGluR or group 1 mGluR (>100 uM); shows anticonvulsant and anxiolytic properties in vivo; orally active.Schizophrenia Phase 2 Clinical.
-
In Vitro——
-
In Vivo——
-
SynonymsEglumegad | Eglumetad
-
PathwayGPCR/G Protein
-
TargetmGluR
-
RecptormGluR
-
Research AreaNeurological Disease
-
IndicationSchizophrenia
Chemical Information
-
CAS Number176199-48-7
-
Formula Weight185.1772
-
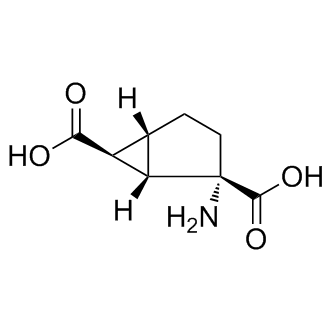
Molecular FormulaC8H11NO4
-
Purity>98% (HPLC)
-
SolubilityDMSO: < 1.9 mg/mL
-
SMILESO=C([C@@]1(N)[C@]2([H])[C@@H](C(O)=O)[C@]2([H])CC1)O
-
Chemical NameBicyclo[3.1.0]hexane-2,6-dicarboxylic acid, 2-amino-, (1S,2S,5R,6S)-
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1. Monn JA, et al. J Med Chem. 1997 Feb 14;40(4):528-37.
2. Menezes MM, et al. Neurosci J. 2013;2013:736439.
3. Profaci CP, et al. Psychopharmacology (Berl). 2011 Jul;216(2):235-43.
molnova catalog



related products
-
AZD8529 mesylate
AZD8529 is a novel potent, selective mGluR2 positive allosteric modulator with EC50 of 195 nM.
-
LY 2812223
A highly potent, functionally selective mGlu2 receptor agonist with EC50 of 13.6 nM in GTPγS functional binding assay.
-
LY-404039
A potent, slective agonist of mGlu2 and mGlu3 receptors with Ki of 149 nM and 92 nM, respectively.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


