BLZ945
CAS No. 953769-46-5
BLZ945( BLZ-945 )
Catalog No. M16814 CAS No. 953769-46-5
BLZ945 (BLZ-945) is a potent, selective, brain-penetrant CSF-1R inhibitor with biochemical IC50 of 1 nM.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 5MG | 65 | In Stock |


|
| 10MG | 102 | In Stock |


|
| 25MG | 158 | In Stock |


|
| 50MG | 205 | In Stock |


|
| 100MG | 357 | In Stock |


|
| 500MG | 822 | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameBLZ945
-
NoteResearch use only, not for human use.
-
Brief DescriptionBLZ945 (BLZ-945) is a potent, selective, brain-penetrant CSF-1R inhibitor with biochemical IC50 of 1 nM.
-
DescriptionBLZ945 (BLZ-945) is a potent, selective, brain-penetrant CSF-1R inhibitor with biochemical IC50 of 1 nM, displays >3,200-fold selectivity over other kinases (c-Kit, PDGFR-β, Flt3, Abl, etc.); inhibits CSF-1-dependent proliferation in bone marrow-derived macrophages (BMDMs) with EC50 of 67 nM, and decreases CSF-1R phosphorylation; blocks glioma progression and significantly improves survival, efficiently limits tumor progression combining with PD-1/PD-L1 blocking antibodies in neuroblastoma.Solid Tumors Phase 2 Clinical(In Vitro):Treatment of bone marrow-derived macrophages (BMDMs) with Sotuletinib inhibits CSF-1-dependent proliferation (EC50=67 nM), and decreases CSF-1R phosphorylation, similar to CSF-1R antibody blockade. Sotuletinib also reduces viability of CRL-2467 microglia, Ink4a/Arf?/? BMDMs (PDG genetic background), and NOD/SCID BMDMs. Importantly, Sotuletinib treatment in culture does not affect proliferation of any PDG-derived tumor cell lines (all Csf-1r-negative), or U-87 MG human glioma cells, and PDG cell tumor sphere formation is unaffected. Thus, Sotuletinib has no direct effects on glioma cells, and perturbs macrophage survival through CSF-1R inhibition. (In Vivo):Mice are treated with Sotuletinib or vehicle, and evaluated for symptom-free survival. Median survival in the vehicle-treated cohort is 5.7 weeks. In striking contrast, Sotuletinib significantly improves long-term survival with 64.3% surviving to the 26-week trial endpoint. This endpoint is chosen because Ink4a/Arf?/? mice develop spontaneous tumors, including lymphomas and sarcomas, beginning at ~30 weeks. Sotuletinib is well-tolerated over long-term treatment, with no visible side-effects, consistent with histopathological studies. Histological grading revealed high-grade, invasive gliomas in all vehicle-treated mice. By contrast, Sotuletinib-treated animals have significantly less-malignant tumors, and no detectable lesions in 55.6% of asymptomatic mice at the endpoint. Mice receiving Sotuletinib shows reduced CSF1R staining in both cervical tumors and the associated stroma, with a significant decrease in CSF1R+ stromal macrophages relative to vehicle-treated mice (P<0.05).
-
In VitroTreatment of bone marrow-derived macrophages (BMDMs) with Sotuletinib inhibits CSF-1-dependent proliferation (EC50=67 nM), and decreases CSF-1R phosphorylation, similar to CSF-1R antibody blockade. Sotuletinib also reduces viability of CRL-2467 microglia, and NOD/SCID BMDMs. Importantly, Sotuletinib treatment in culture does not affect proliferation of any PDG-derived tumor cell lines (all Csf-1r-negative), or U-87 MG human glioma cells, and PDG cell tumor sphere formation is unaffected. Thus, Sotuletinib has no direct effects on glioma cells, and perturbs macrophage survival through CSF-1R inhibition.
-
In VivoMice are treated with Sotuletinib or vehicle, and evaluated for symptom-free survival. Median survival in the vehicle-treated cohort is 5.7 weeks. In striking contrast, Sotuletinib significantly improves long-term survival. This endpoint is chosen because Ink4a/Arf?/? mice develop spontaneous tumors, including lymphomas and sarcomas, beginning at ~30 weeks. Sotuletinib is well-tolerated over long-term treatment, with no visible side-effects, consistent with histopathological studies. Histological grading revealed high-grade, invasive gliomas in all vehicle-treated mice. By contrast, Sotuletinib-treated animals have significantly less-malignant tumors, and no detectable lesions in 55.6% of asymptomatic mice at the endpoint. Mice receiving Sotuletinib shows reduced CSF1R staining in both cervical tumors and the associated stroma, with a significant decrease in CSF1R+ stromal macrophages relative to vehicle-treated mice (P<0.05).
-
SynonymsBLZ-945
-
PathwayTyrosine Kinase
-
TargetCSF1R
-
RecptorCSF-1R
-
Research AreaCancer
-
IndicationSolid Tumors
Chemical Information
-
CAS Number953769-46-5
-
Formula Weight398.4787
-
Molecular FormulaC20H22N4O3S
-
Purity>98% (HPLC)
-
SolubilityDMSO: ≥ 30 mg/mL
-
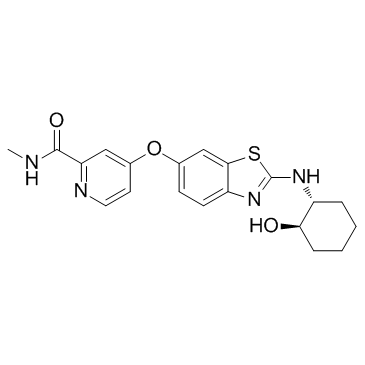
SMILESO=C(NC)C1=NC=CC(OC2=CC3=C(N=C(N[C@@H]4CCCC[C@H]4O)S3)C=C2)=C1
-
Chemical Name2-Pyridinecarboxamide, 4-[[2-[[(1R,2R)-2-hydroxycyclohexyl]amino]-6-benzothiazolyl]oxy]-N-methyl-
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1. Pyonteck SM, et al. Nat Med. 2013 Oct;19(10):1264-72.
2. Pass HI, et al. Oncotarget. 2016 Aug 30;7(35):56408-56421.
3. Mao Y, et al. Clin Cancer Res. 2016 Aug 1;22(15):3849-59.
4. Quail DF, et al. Science. 2016 May 20;352(6288):aad3018.
molnova catalog



related products
-
ARRY-382
ARRY-382 is a highly selective oral inhibitor of CSF-1R with an IC50 of 9 nM.
-
c-Fms-IN-13
c-Fms-IN-13 (compound 14) is a potent FMS kinase inhibitor (IC50 = 17 nM) and serves as an anti-inflammatory agent.
-
Axatilimab
Axatilimab (SNDX-6352) is a human IgG4 antibody targeting the colony-stimulating factor 1 receptor (CSF-1R), which can be used for the study of immune system disorders and respiratory diseases.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


