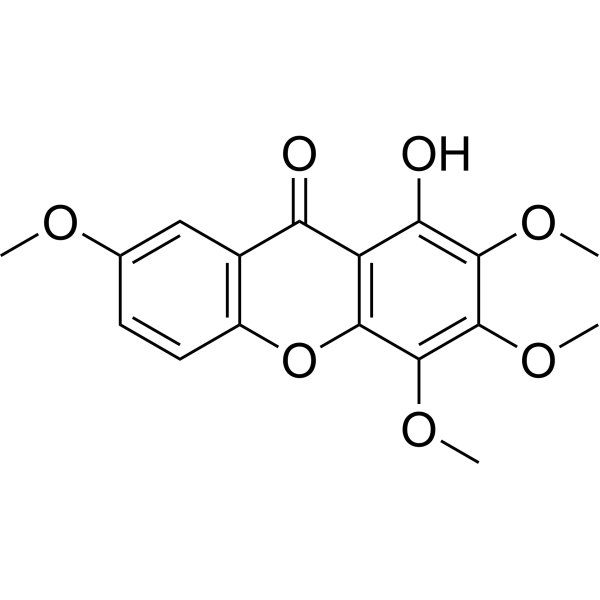
1-Hydroxy-2,3,4,7-tetramethoxyxanthone
CAS No. 14103-09-4
1-Hydroxy-2,3,4,7-tetramethoxyxanthone( —— )
Catalog No. M31474 CAS No. 14103-09-4
1-Hydroxy-2,3,4,7-tetramethoxy-xanthone has vasodilatory action, it can cause vasodilation in the coronary artery pre-contracted with 1uM 5-hydroxytryptamine (5-HT), with the EC 50 value of 6.6±1.4 uM.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 5MG | 414 | In Stock |


|
| 50MG | Get Quote | In Stock |


|
| 100MG | Get Quote | In Stock |


|
Biological Information
-
Product Name1-Hydroxy-2,3,4,7-tetramethoxyxanthone
-
NoteResearch use only, not for human use.
-
Brief Description1-Hydroxy-2,3,4,7-tetramethoxy-xanthone has vasodilatory action, it can cause vasodilation in the coronary artery pre-contracted with 1uM 5-hydroxytryptamine (5-HT), with the EC 50 value of 6.6±1.4 uM.
-
Description1-Hydroxy-2,3,4,7-tetramethoxy-xanthone has vasodilatory action, it can cause vasodilation in the coronary artery pre-contracted with 1uM 5-hydroxytryptamine (5-HT), with the EC 50 value of 6.6±1.4 uM. 1-Hydroxy-2,3,4,7-tetramethoxyxanthone can effectively inhibit the osteoclast differentiation in a co-culture system with mouse osteoblastic calvarial cells and bone marrow cells.
-
In Vitro——
-
In Vivo——
-
Synonyms——
-
PathwayOthers
-
TargetOther Targets
-
Recptor——
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number14103-09-4
-
Formula Weight332.3
-
Molecular FormulaC17H16O7
-
Purity>98% (HPLC)
-
Solubility——
-
SMILES——
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
molnova catalog



related products
-
DO-264
DO-264 is an inhibitor of autohydrolase structural domain 12 (ABHD12) (IC50: 11 nM) and an inhibitor of cellular LysoPS degradation that enhances LPS-induced phagocytosis.
-
Ermanin
Ermanin is a flavonoid isolated from Tanacetum microphyllum.Ermanin inhibits platelet aggregation and has anti-tuberculosis and anti-viral/bacterial properties.
-
Rubinaphthin A
Rubinaphthin A is a natural product from Rubia cordifolia.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


